|
Protons belong to the family of particles known as hadrons. Proton A subatomic particle that is one of the basic building blocks of the atoms that make up matter. Recommendations for the Naming of Elements of Atomic Numbers Greater than 100. You can identify an element by its atomic number, so in this case, it would be carbon C. 
Neutrons belong to the family of particles known as hadrons. The atomic number is the number of protons in the nucleus. Jump to main content Periodic Table Home History Alchemy Podcast Video Trends Periodic Table Home History Alchemy Podcast Video Trends You do not have JavaScript enabled. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Neutron A subatomic particle carrying no electric charge that is one of the basic pieces of matter. Element Boron (B), Group 13, Atomic Number 5, p-block, Mass 10.81. Examples include hydrogen, oxygen, carbon, lithium and uranium. Chemical also can be an adjective to describe properties of materials that are the result of various reactions between different compounds.Įlectron A negatively charged particle, usually found orbiting the outer regions of an atom also, the carrier of electricity within solids.Įlement (in chemistry) Each of more than one hundred substances for which the smallest unit of each is a single atom. For example, water is a chemical made when two hydrogen atoms bond to one oxygen atom. The nucleus is orbited by a cloud of negatively charged electrons.Ītomic Having to do with atoms, the smallest possible unit that makes up a chemical element.Ītomic number The number of protons in an atomic nucleus, which determines the type of atom and how it behaves.Īverage (in science) A term for the arithmetic mean, which is the sum of a group of numbers that is then divided by the size of the group.Ĭhemical A substance formed from two or more atoms that unite (bond) in a fixed proportion and structure. Atoms are made up of a dense nucleus that contains positively charged protons and uncharged neutrons. The year 2019 marks the 150th anniversary of the periodic table, first conceived in 1869.Ĭheck out the full list of Scientists Say.Ītom The basic unit of a chemical element. That brought the number of known elements to 118 and completed the 7th row of the table. Starting in the 1940s, scientists found many new elements by colliding atoms or pieces of atoms.Īt the end of 2018, chemists confirmed four elements that had never been observed before. This is a group of elements such as helium that don’t like to react with other elements. These include the noble gases, identified in 1890. The periodic table continued to grow as scientists discovered more elements. Other scientists were working on their own periodic tables, but Mendeleev published his table first. He saw trends in the elements’ properties that varied over specific intervals, or periods. In 1869, the Russian chemist Dimitri Mendeleev lined up the 63 known elements in order by their atomic weights. An atomic weight is the average weight of one atom of an element. But they did understand that elements had different atomic weights. Back then, scientists didn’t know about the protons, neutrons and electrons that make up atoms. It also shows how its electrons are arranged.ĭuring the mid-1800s, many chemists looked for patterns that explained how elements interacted. This spelling is encountered in many references. An element’s place also shows how likely it is to react. The original German spelling of the name of the 1901 Nobel Prize in Physics Laureate was Wilhelm Conrad Röntgen. The first thing to do is to get a list of the elements 1 Hydrogen (H) 2 Helium (He) 3 Lithium (Li) 4 Beryllium (Be) 5 Boron (B) 6 Carbon (C) 7 Nitrogen (. Those on top of the chart have the fewest protons. First, the elements are organized by atomic number, or how many protons they have. The location of each square in the table tells many things about each element. A square contains one or two letters that stand for the element’s name, and numbers that tell about that element’s properties. The table is made up of over a hundred squares. This is a chart that shows all the known chemical elements. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.Periodic table (noun, “peer-ee-AHH-dik TAY-bul”) Please note that the elements do not show their natural relation towards each other as in the Periodic system. The first chemical element is Hydrogen and the last is Ununoctium.  
The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. This list contains the 118 elements of chemistry. 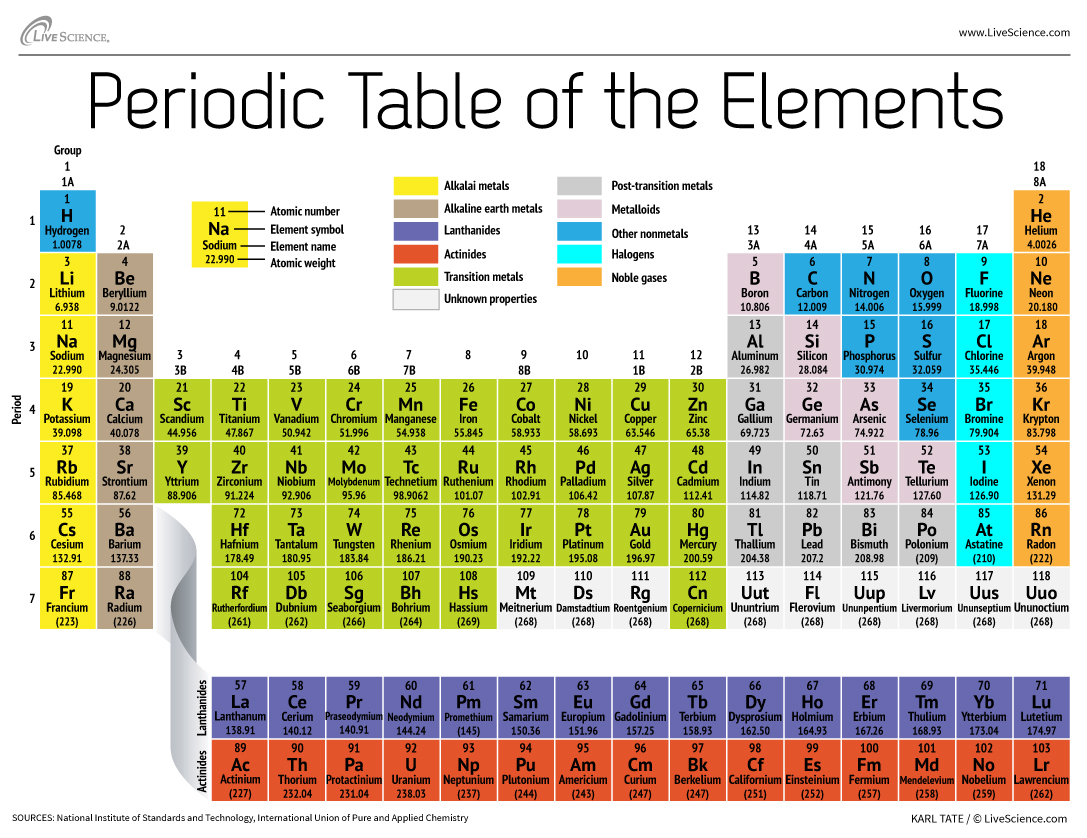
Plant Inspection & Process OptimalisationĬhemical elements listed by atomic number The elemenents of the periodic table sorted by atomic numberĬlick on any element's name for further chemical properties, environmental data or health effects.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
